 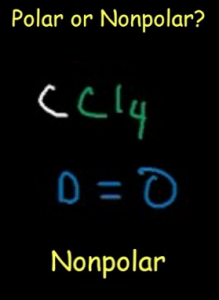
Due to this symmetry, tetrachloromethane is said to be non-polar, even though it contains polar bonds. In case of CCl4, the charges are distributed uniformly around the central carbon atom. Molecules having their charges unevenly distributed are said to be polar in nature, whereas, those molecules whose charges are evenly distributed are non-polar. When it comes to polarity of a compound, we must understand the fact that it is directly related to its structure and shape. The atom of each element present in this compound is stable, in turn, making the compound quite stable. Identify the Central Atom Recognize that carbon will be the central atom. Carbon (group 14) brings four valence electrons, and there are four chlorine atoms (group 17), each contributing seven valence electrons. Considering the four atoms of chlorine, each atom will have 6 electrons around it. Count Valence Electrons Begin by determining the total number of valence electrons. The remaining 24 atoms must be distributed in the structure in such a way that all the atoms have 8 electrons in their valence orbital. The number of electrons around the carbon atom is 8. Total number of electrons in the compound = Total number of valence electrons of (carbon + chlorine) Total number of electrons in the compound = (1 X 4) + (4 X 7) = (4 + 28) = 32 Is the lewis structure of NCl3 the same as NF3 and. according to the VSEPR chart, the electron geometry of NCl3 is tetrahedral while molecular geometry is trigonal pyramidal. It has a melting point of 40☌ and a boiling point of 71☌. The carbon atom shares 1 electron each to form one covalent bond with every chlorine atom to achieve its octet. It is not soluble in water but soluble in benzene, PCl3, CCl4, etc. This implies that carbon requires 4 electrons and chlorine requires 1 electron to satisfy its octet. Thus, carbon and chlorine have 4 and 7 valence electrons, respectively. 
When combined with a halogen, it forms a compound. (In the 3rd blank type whether it is a polar, non-polar or ionic compound) 2 In its reaction. 
We will learn how to draw the lewis structure of CCl 4 step by step in this tutorial. Carbon atom is the center atom and each chlorine atom has 3 lone pairs. Lewis structure of CCl 4 contains four C-Cl bonds. There are four chlorine atoms and one carbon atom in CCl 4 molecule. As we already know, the atomic number of carbon atom is 6, and that of chlorine is 17. Question: The electron domain geometry of CCl4 is and its molecular geometry is It is a compound. Carbon tetrachloride (CCl 4) is an alkyl halide compound. Carbon forms one covalent bond with each chlorine atom, which is electronegative in nature. Identify the Central Atom Recognize that carbon will be the central atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
